High-Dose Vitamin D Supplements Significantly Reduce ICU Admissions, COVID-19 Deaths – OpEd
By IPS
By Sunil J. Wimalawansa*
There is considerable evidence that vitamin D reduces the risk and severity of COVID-19 (Mercola 2020 ; Wimalawansa, 2020). More than 50 clinical studies have published confirming that high doses of vitamin D administered early in persons with COVID-19 significantly reduce complications and the need for ICU admissions.
Additionally, five randomised controlled clinical trials (RCTs) were published using high-dose vitamin D in COVID-19 patients, and over 20 other large RCTs are ongoing. To obtain benefits, however, doses high enough must be administered early in the disease.
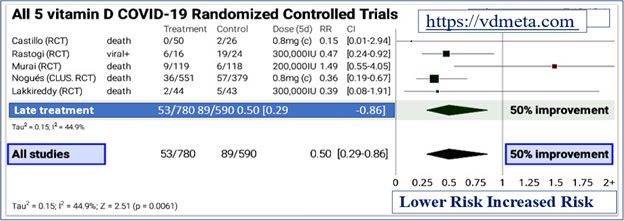
Among others, few examples of countries where these RCTs conducted were Brazil (Murai et al, 2021), India (Rastogi, et al, 2020 ; Lakkireddy, 2021), and Spain (Castillo, et al, 2020 ; Nogués et al, 2021). The vitamin D3 doses used were between 130,000 IU to 300,000 IU or the equivalent of 25-hydroxyvitamin D [25(OH)D, also known as calcifediol] as used by Castillo et al. in Spain.
As shown in the meta-analysis, figures 1 and 2, at https://vdmeta.com, even with slightly less optimal doses of calcifediol resulted in over 50% (95% confidence interval, 14 to 71%) reduction in severe adverse outcomes by preventing the development of cytokine storm and death.
The effect of early treatment with high-dose vitamin D in persons with COVID-19 illustrated in Figure 1 (adapted from https://vdmeta.com).
There are no adverse effects of high-dose vitamin D3 supplementation of up to 600,000 IU administered as single or divided doses. Also, long-term supplementation of 50,000 IU/day vitamin D3 have not reported adverse effects (McCullough, et al, 2019).
An example of cost-effectiveness of treating persons with “Symptomatic” COVID-19, using high dose vitamin D.
In examples below, the expected death rates is adjustable with 75% reduction following vitamin D; so as the cost of vitamin D per patient:
Conclusion
Vitamin D, 200,000 IU dose in single or divided doses is safe and highly cost-effective in reducing complications and deaths in persons with SARS-CoV-2 infection. Early use of vitamin D (100,000 to 600,000 IU) can reduce ICU admissions by ~80%. Therefore, vitamin D can greatly reduce the need for expanding ICU beds AND cost less than 0.001% of using an ICU bed/day.
Additional information regarding bolus D3 and oral calcifediol:
There is little vitamin D3 (cholecalciferol) in food and multivitamins, including fortified food. Without passing through glass or sunscreen, skin exposure to ultraviolet-B from high-elevation sunlight can produce sufficient D3 for bone and immune system health.
However, low elevation sunlight in winter, clouds, pollution, clothing cover, melanin-rich skin and concerns about skin cancer mean that most people are vitamin D deficient unless they take vitamin D supplements.
Conversion of D3 in the liver to the circulating form, 25(OH)D, and for the immune cells to function takes two to five days. 25(OH)D is measured in the blood test and has a half-life of ~three weeks. Recent research shows that the immune system requires at least 40 ng/ml (100 nmol/L) circulating 25(OH)D to support autocrine (inside each cell) and paracrine (to nearby cells) signalling. Please refer to the research articles at: https://vitamindstopscovid.info/02-autocrine/.
In the long term, such levels can be attained, on average for 70 kg adults, with 0.125 mg (5,000 IU) D3 per day. This is 72 IU per kg body weight per day and 100 IU/kg for people suffering from obesity due to fat absorption of 25(OH)D.
The link to research articles cited follows: https://vitamindstopscovid.info/01-supp/.
Since most people-and almost all who are at risk of severe COVID-19 symptoms-have circulating 25(OH)D levels much lower than 40 ng/mL, such as between 5 and 25 ng/mL, their D3 supplementation should begin at 5 to 10 times the long-term quantity of 4,000 IU/day needed, in order to attain and maintain the required blood 25(OH)D levels rapidly.
While vaccines markedly reduce the severity of and deaths from COVID-19, vitamin D cost less than 1% of a COVID vaccine.
As reported in several studies, most hospitalised patients with COVID-19 can be assumed to be vitamin D deficient. Their health and survival depend mainly on raising their 25(OH)D levels to at least 40 ng/ml, ideally within hours, rather than days or weeks.
Some of the trials mentioned above attempted to do this with bolus D3 doses, except that in the Murai et al. trial, given too little treatment, too late (i.e., faulty study design).
The urgency of 25(OH) D repletion is acute for all those suffering from, or at risk of, hyper-inflammatory immune dysregulation (i.e., cytokine storm), which causes severe COVID-19 and death.
The primary mechanism of this recently elucidated by McGregor, et al, 2020-Th1 regulatory lymphocytes remain stuck in their initial pro-inflammatory program and fail to switch to their anti-inflammatory shutdown program due solely to lack of 25(OH)D.
This explains the extraordinary success of trial by Castillo et al., in which patients were given 0.532 mg oral calcifediol, which raises circulating 25(OH)D levels to about 50 ng/ml in four hours (Sune Negre, 2016).
This resulted in the reduction of ICU admissions by 90% (from 50% to 2%) and deaths from 8% to zero. The earlier the treatment is given, the better the clinical outcomes are.
Calcifediol has been hard to obtain but is now available in the USA and Canada without prescription: https://dvelopimmunity.com. Sixty 20 microgram tablets for $30 contain 1.2 mg calcifediol-more than twice the single initial dose used by Castillo et al.
In that trial, 0.266 mg calcifediol was also given on days 3, 7, 14, etc. However, maintenance of the initial boost to 25(OH)D can most easily be achieved with 5,000 to 10,000 IU D3 per day.
We have urged governments of Sri Lanka and India, in particular, to seriously consider obtaining calcifediol tablets to prevent severe complications, ICU admissions, and deaths from COVID-19, which is highly cost-effective.
*Sunil J. Wimalawansa is a Professor of Medicine, Endocrinology & Nutrition, Director CardioMetabolic Institute, USAContact information: *Sunil J. Wimalawansa, MD, PhD, MBA, DSc [email protected]