Taking A Deep Look Into Animals
A collaborative project driven by researchers of the Max Perutz Labs Vienna, a joint venture of the University of Vienna and the Medical University of Vienna, and the TU Wien (Vienna) allows researchers to look deep into organs and nervous systems of animals, ranging from squids and worms to fish and salamanders.
Analyses of individual cells in the context of whole organs or tissues is becoming increasingly important in biology. A standard approach so far was to cut larger tissues into thin layers, study each of these sections, and then piece the information again together into a 3D model. However, this is a laborious process, and often yields incomplete results. For instance, the cells that make up our nervous systems possess long extensions that can reach through the entire body. Reconstructing such projections from small slices is extremely challenging.
An elegant solution to avoid this is provided by tissue clearing techniques that can render opaque tissues transparent. When applied to complex tissues, including the brain, such techniques allow to visualize individual cells and their extensions, enabling scientists to capture 3D images of cells and tissues without the need of sectioning. However, existing clearing techniques so far were not optimized to remove a variety of pigments that are present in tissues, and that limit how deeply these specimens can be imaged. Therefore, despite the power of tissue clearing approaches, they have essentially remained restricted to specific unpigmented organs like the brain, and a handful of model species that have reduced pigmentation.
In a team effort, researchers from the Max Perutz Labs, the Medical University of Vienna and the TU Wien (Vienna) and their collaborators have now developed a new method that combines tissue clearing with the removal of various pigment types as they are characteristic for most animals. This new approach – dubbed “DEEP-Clear” – has now been published in the international journal Science Advances.
A toolkit for imaging biomolecules in the nervous systems of a broad panel of species
An important observation that helped to develop the new method was that the combination of different chemical treatments had a synergistic effect, allowing for fast depigmentation and tissue clearing.
“Shortening chemical processing preserves the integrity of tissues and organisms, so that the molecules and internal structures of interest are more likely to be retained”, explains Marko Pende, the developer of the clearing method, from the lab of Hans-Ulrich Dodt at the TU Wien and the Center for Brain Research (CBR) of the Medical University of Vienna, and one of the first authors of the study. This way multiple organisms could be imaged from different clades ranging from mollusks to bony fish to amphibians.
“These are just a few examples. We believe that the method is applicable to multiple organisms. It was just not tried yet”, explains Prof. Hans Ulrich Dodt, senior author of the study.
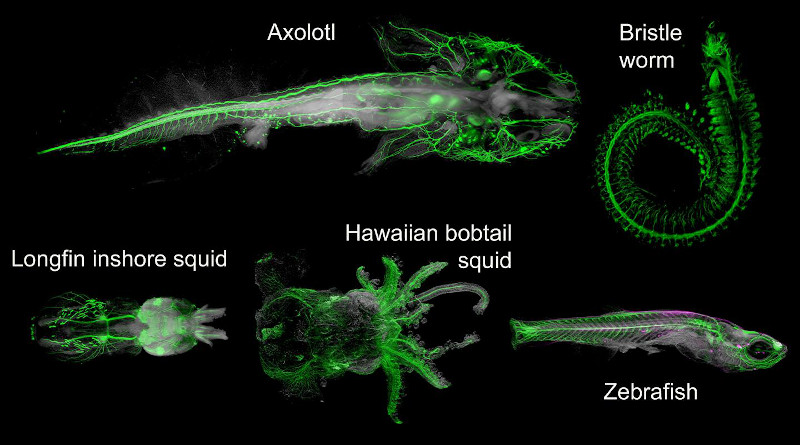
The team then systematically explored which types of molecules could still be marked and detected in DEEP-Clear-processed samples, investigating species ranging from squids and worms to fish and salamanders. This work – largely performed by PhD student Karim Vadiwala in the lab of Florian Raible at the Max Perutz labs – showed that DEEP-Clear was compatible with the detection with a variety of important biomolecules, allowing to image specific proteins, DNA markers and RNA in intact specimens.
“This versatility of DEEP-Clear makes it a highly attractive tool to explore a range of animals for which standard tissue clearing techniques currently would not be sufficient”, explains Karim Vadiwala (Max Perutz Labs).
A 3D view of whole animal nervous systems to explore neural stem cell biology
Besides its compatibility with many species, another attractive feature of DEEP-Clear is that the transparency of the processed organisms allows to image samples across scales: On the one hand, the team looked into very small details such as contact points between neurons, or individual clusters of dividing cells. On the other hand, they took advantage of the latest generation of so-called light-sheet microscopes developed by the Dodt lab, in which two-dimensional laser light is used to rapidly scan a whole sample, resulting in a full three-dimensional model created on the computer.
“Using a very thin light-sheet enables us to overcome a lot of optical limitations and allows us to generate such high-resolution images, even from samples that are several millimeters thick”, says the designer of the microscopes, Dr. Saiedeh Saghafi (TU Wien).
The expectation of the team is that “DEEP-Clear” will serve to popularize tissue clearing, allowing researchers around the world to intensify molecular and cellular research in a variety of species that exhibit highly interesting, but poorly explored neuroscientific features. For instance, worms, fish, and salamanders can regenerate parts of their central nervous system, suggesting that they possess molecular capacities that humans and other mammals have lost.
“Visualizing the responsible stem cells, and investigating their molecular make-up, or their contribution to regenerated tissue, will be greatly facilitated by DEEP-Clear”, believes Dr. Florian Raible (Max Perutz Labs), who coordinated the study that also involved additional researchers in the groups of Dr. Oleg Simakov (Uni Vienna) and Dr. Elly Tanaka (Institute for Molecular Pathology).