Novel Coronavirus 2019: Global Implications And Responses – Analysis
By CRS
By Tiaji Salaam-Blyther, Susan V. Lawrence and Sara M. Tharakan*
On December 31, 2019, China’s government notified the World Health Organization (WHO) of patients with pneumonia of unknown cause in Wuhan, capital of the central Chinese province of Hubei. On January 7, 2020, Chinese scientists isolated a previously unknown coronavirus in the patients, and on January 12, the scientists made its genetic sequence available to WHO and international partners. WHO initially named the virus 2019- nCoV and later renamed it severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The virus can cause a disease in humans that WHO is calling coronavirus disease 2019 (COVID-19).
The Virus
Coronaviruses are a large family of zoonotic viruses (viruses transmissible between animals and humans) that can cause illness ranging from the common cold to more severe diseases such as Middle-East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome (SARS). The most common symptoms among confirmed COVID-19 patients include fever, dry cough, and shortness of breath.
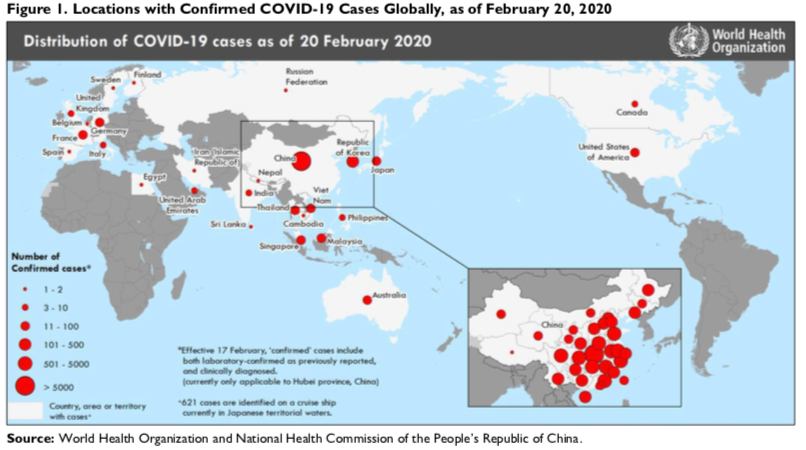
As of February 20, 2020, WHO reported more than 75,000 confirmed COVID-19 cases globally, including more than 2,100 deaths. Approximately 98% of the cases and all but 11 of the deaths were in mainland China. More than 80% of all cases in mainland China were reported in Hubei Province, the outbreak epicenter.
Outside of mainland China, the jurisdictions with the highest reported caseloads were South Korea (104), Japan (85), Singapore (84), Hong Kong (65), Thailand (35), Taiwan (24), Malaysia (22), Vietnam (16), Germany (16), Australia (15), and the United States (15). WHO reported that over 600 cases, more than half the total COVID-19 cases outside mainland China, were detected from the Diamond Princess cruise ship, docked alongside Yokohama, Japan. One passenger from the Westerdam cruise ship, docked alongside Sihanoukville, Cambodia, has also tested positive for the virus.
Within days of learning the genetic sequence of the virus, scientists around the globe developed lab tests to diagnose COVID-19. Current diagnostic supplies are insufficient to meet global demand, however. While trying to increase supply of these tests, developers are also trying to create faster, cheaper, and more easily administered tests.
No specific treatments or vaccines for COVID-19 exist, although WHO has developed a disease commodity package that lists essential biomedical equipment, medicines, and supplies health workers need to care for patients with the disease. The U.S. National Institutes of Health (NIH) indicated in late January that a candidate vaccine for COVID-19 could be ready for early-stage human testing within three months, compared to the 20 months it took to begin early-state development of an investigational SARS vaccine.
Developments in China
In 2002-2003, China’s government was widely criticized for waiting three months to report the outbreak of SARS to WHO and its own people. In the case of COVID-19, China’s reporting to WHO came earlier in the outbreak, but the government did not raise the alarm to its own people until three weeks later, on January 20, 2020.
Chinese authorities appear to have sought to suppress certain information about the outbreak in the interim, including by disciplining eight medical workers for “rumor mongering” after they shared information about the pneumonia cases with their social circles. One of those medical workers, ophthalmologist Li Wenliang, 33, later died from COVID- 19. Observers have suggested that by withholding information, the government may have squandered an early window of opportunity to stem the virus’s spread.
After January 20, Chinese authorities began taking aggressive actions to prevent the virus from spreading further. By February 20, they had imposed cordon sanitaires, or restrictions on movement in and out of defined areas, on as many as 760 million people and confined as many as 150 million people to their homes, according to a New York Times analysis.
Arguing that such measures should be “strictly necessary to achieve a legitimate objective,” U.S.-based Human Rights Watch suggested that China’s January 23 lockdown of Wuhan “was arbitrary and potentially discriminatory.” WHO Director-General Tedros Adhanom Ghebreyesus has praised China, however, for the “extraordinary measures it has taken to contain the outbreak, despite the severe social and economic impact those measures are having on the Chinese people.” (See also CRS In Focus IF11434, The Coronavirus: U.S.-China Economic Considerations.)
With positive trends in recent official case data in China, China’s leaders have suggested that China may soon be able to declare victory over the virus and get the country back to work. Those data trends include declines in the number of daily new cases outside Hubei Province since February 4, and in Hubei Province outside Wuhan since February 13. On February 17, a top-level Communist Party committee formed to manage the outbreak response concluded, “The possibility of a wider epidemic has been avoided.”
Weighed against that optimism are widespread questions about the integrity of China’s data; a worrying rise in infections in Beijing; reports of continued shortages of supplies at hospitals in Hubei; reports of shortages of test kits elsewhere in China; and morale-sapping reports of the deaths of more high-profile patients, including a Wuhan hospital director, Liu Zhiming, on February 18.
WHO Response
PHEIC. Following the emergence of a disease that might be deemed a Public Health Emergency of International Concern (PHEIC), WHO convenes an advisory group, known as the IHR Emergency Committee, to review data and make recommendations to the WHO Director-General related to the disease.
On January 22, at its initial meeting, WHO’s 15-member IHR Emergency Committee for Pneumonia due to the Novel Coronavirus COVID-19 agreed on the “urgency of the situation.” At a second meeting on January 30, the committee, which includes Dr. Martin Cetron of the U.S. Centers for Disease Control and Prevention (CDC), concluded the outbreak had become a PHEIC.
The same day, WHO Director-General Tedros issued a PHEIC declaration, prompting countries to take specific actions, including heightening surveillance and reporting of the disease. In the case of the United States, Department of Health and Human Services (HHS) Secretary Alex Azar declared “a public health emergency for the entire United States.”
A PHEIC declaration can also prompt countries to provide additional resources for global and domestic response and enable WHO to access certain emergency funding, such as from the United Nations Central Emergency Response Fund (CERF) and the World Bank Pandemic Emergency Financing Facility (PEF).
WHO-Led Expert Mission. On January 28, four weeks after China first informed WHO of the outbreak, Chinese leader Xi Jinping agreed to accept a WHO-led expert mission to review data and conduct field visits, with the goal of informing COVID-19 response in China and globally. International mission members arrived in China on February 15 and 16. They include experts from CDC and NIH and institutions in Germany, Japan, Nigeria, Russia, Singapore, and South Korea. The mission is to visit Beijing and two provinces, but not Hubei.
WHO Funds. On February 5, WHO announced a $675 million COVID-19 preparedness plan for February through April. It aims to provide international coordination and operational support, bolster country readiness and response capacity (particularly in low-resource countries), and accelerate relevant research and innovation. As of February 19, donors had pledged to provide $26.0 million in support of the plan and WHO had received $1.2 million.
U.S. Response
Funds for Global COVID-19 Control. On January 29, President Donald J. Trump announced the formation of the President’s Coronavirus Task Force, led by HHS and coordinated by the National Security Council. On February 7, the Department of State and the U.S. Agency for International Development committed to use up to $100 million of existing funds to assist China and other affected countries with COVID-19 responses. HHS has made available up to $105 million from the Infectious Disease Rapid Response Reserve Fund for domestic and international COVID-19 responses.
Travel Issues. The Department of Homeland Security has issued instructions to quarantine travelers returning to the United States after stays in China and to bar entry to most foreigners with recent travel to China. The State Department has issued a Level 4 (“Do Not Travel”) advisory for China, and evacuated more than 800 U.S. citizens and others from Wuhan. The Department ordered the mandatory departure of nonemergency U.S. personnel and their families from the U.S. consulate in Wuhan. For other U.S. diplomatic posts in China, the Department authorized the voluntary departure of nonemergency U.S. personnel and their families. China has sharply criticized all those measures, accusing the United States of having “inappropriately overreacted.”
*About the authors: Tiaji Salaam-Blyther, Specialist in Global Health Susan V. Lawrence, Specialist in Asian Affairs Sara M. Tharakan, Analyst in Global Health and International Development
Source: This article was published by Congressional Research Service (PDF)

